Disease Areas of Focus:
CPD100 in ophthalmology for wet age-related macular degeneration
CPD100Li in oncology for solid tumors
Cascade Prodrug was founded out of technology developed originally at University of Oregon and Oregon State University focused on formulations of novel hypoxia-activated prodrugs of vinca alkaloids.
Cascade has designed CPD100, a prodrug that contains a modified (oxidized) form of vinblastine (vinblastine-N-oxide) which becomes activated under a hypoxic environment. Upon delivery to hypoxic tissue environments, CPD100 (vinblastine-N-oxide), will be reduced to the active vinblastine moiety. This approach is expected to result in therapeutic concentrations of vinblastine at the hypoxic regions of the targeted tissues.

CPD100 is under development in two separate formulations, each designed for a specific route of administration and clinical use:
- CPD100 is being developed for patients with wet age‑related macular degeneration and will be delivered through suprachoroidal injection.
- CPD100Li, is a liposomal formulation of CPD100 with enhanced pharmacokinetic properties, designed for intravenous administration and intended for development in patients with solid tumors
WET AGE-RELATED MACULAR DEGENERATION (AMD)

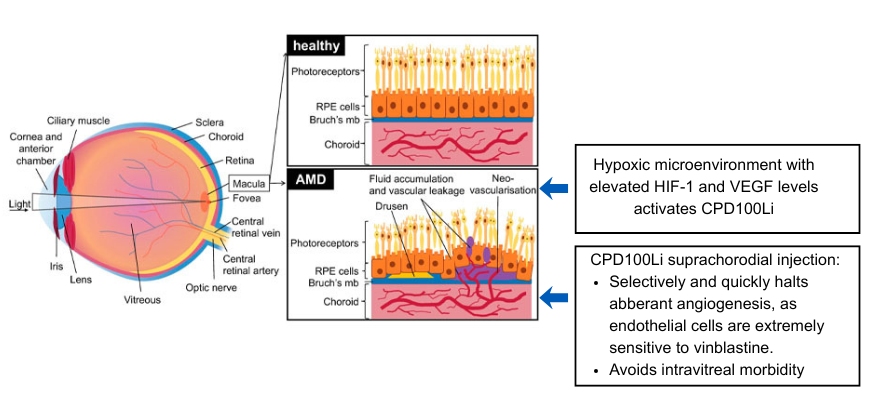
Wet age-related macular degeneration (AMD) is a hypoxia-driven retinal disease characterized by progressive macular damage and the development of abnormal choroidal blood vessels beneath the retina (choroidal neovascularization, CNV). These fragile vessels leak fluid and blood, triggering inflammation, hemorrhage, and fibrotic remodeling of the macula that ultimately leads to irreversible central vision loss.
Current standard-of-care therapy targets the principal mediator of pathologic angiogenesis—vascular endothelial growth factor (VEGF)—using intravitreal anti-VEGF agents. While these therapies can suppress CNV activity, they are not curative and require ongoing, repeated injections to maintain disease control.
RATIONALE OF USING CPD100 IN WET AMD
CPD100 is a hypoxia-activated therapy (HAT) engineered to remain inert under normoxic conditions and become selectively activated in hypoxic tissue, where it releases vinblastine or related cytotoxic species. Vinblastine is a well-characterized inhibitor of microtubule assembly that induces cell-cycle arrest and cell death. Preclinical studies demonstrate that very low concentrations of vinblastine suppress the proliferation of endothelial cells under hypoxia while sparing retinal cells under the same conditions. Both CPD100 and vinblastine have also been shown to reduce VEGF-producing M2 macrophages across multiple models. Together, these properties suggest that CPD100 can selectively target proliferating endothelial cells and VEGF-producing cells that drive choroidal neovascularization in the hypoxic macula, while minimizing toxicity to quiescent retinal cells.
Solid Tumors
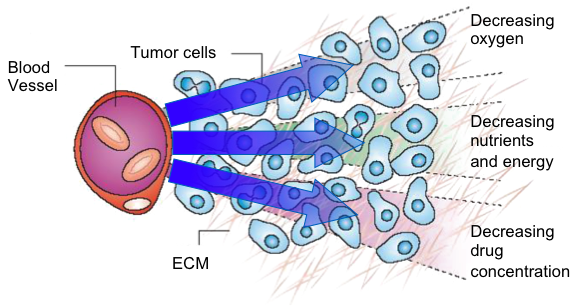
As solid tumors grow, they develop disorganized vasculature that creates regions of low oxygen (hypoxia) and poor nutrient supply. Hypoxic regions within solid tumors drive extensive biological disruption, including compromised genome integrity, heightened chromosomal instability, increased mutagenesis, reduced immune surveillance, and uneven perfusion across the tumor microenvironment. These regions also have slow growing cell populations, hallmarks of solid tumors, and difficult to reach and treat with current standards of care. These pressures accelerate the emergence of invasive and metastatic clones and contribute to resistance against local therapies (surgery, radiotherapy) as well as systemic treatments (chemotherapy, targeted agents, and immunotherapies).
Cascade Prodrug’s strategy is to exploit the hypoxic tumor microenvironment, enabling selective activation of CPD100Li within hypoxic cell populations—either as a monotherapy or in combination with standard chemotherapies or immune checkpoint inhibitors.
RATIONALE OF USING CPD100Li IN SOLID TUMORS
CPD100Li is a liposomal formulation of CPD100 designed for systemic intravenous administration. The liposomal encapsulation significantly extends the circulating half-life of the prodrug compared with vinblastine or non‑liposomal CPD100, enabling greater tumor exposure, less frequent dosing, and an improved safety profile.
Within hypoxic tumor regions, CPD100Li is preferentially activated to regenerate vinblastine, a well‑characterized inhibitor of microtubule assembly that induces cell‑cycle arrest and apoptosis in proliferating tumor cells. Because vinblastine is widely used across solid‑tumor chemotherapy regimens and as monotherapy for pediatric low‑grade glioma, CPD100Li has the potential to improve its therapeutic index through selective hypoxia‑based activation.
Both CPD100 and vinblastine have also been shown to enhance immunosurveillance by reducing immunosuppressive M2 macrophages across multiple tumor models, offering potential synergy with immunotherapies.
Together, these features position CPD100Li to deliver improved safety, enhanced efficacy, and deeper tumor targeting in hypoxia‑prone solid tumors.

